 Metal displacement occurs when a more reactive metal forms a cation and displaces a less reactive metal from its compounds.ĥ. Reactivity of metals and non-metals The pattern of reactivity of the group 1 elements and the group 7 elements shows the general pattern for the reactivity of other groups. This provides the driving force for metal displacement reaction. In other words, a highly reactive metal prefers to be a cation while its less reactive friend prefers to be a free element. A less reactive metal is a hoarder - it would rather have its valence electrons. The reactivity series tells us how much a metal wants to be a cationĪ more reactive metal donates electrons more readily to form a cation. More reactive metals react more vigorously, increasing the rate of effervescence of hydrogen gas.Ĥ. Secondly, reactivity affects the rate of reaction. The unreactive metals do not react at all, even when we use strong acid or high temperature. 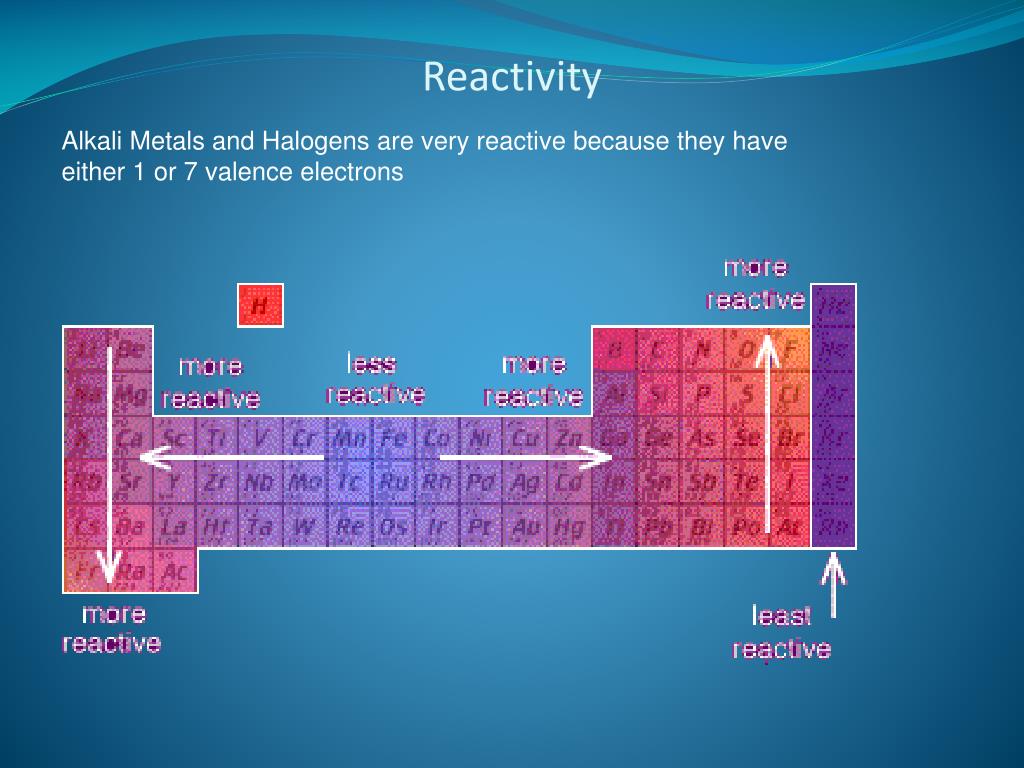
Moderately reactive metals require a higher temperature to react with water.Highly reactive metals are trigger happy, reacting with acids and even water at room temperature.Reactive metal + hydrochloric acid ⟶ metal chloride + hydrogenįirstly, reactivity affects what a metal can react with. Reactive metal + steam ⟶ metal oxide + hydrogen Reactive metal + water ⟶ metal hydroxide + hydrogen We need oxygen all the time throughout our lives. The group 16 elements are intimately related to life. These can be found in nature in both free and combined states. The reactivity series tells us if a reaction will happen and under what conditions Metal It consists of the elements oxygen, sulfur, selenium, tellurium and polonium. The trio sit in the same column within the transition metal hood.ģ. The Trio of Unreactive MetalsĪnd the least reactive metals we need to know are copper, silver, and gold. Lead in Group IV is also moderately reactive. Transition metals are generally less reactive than the main group metals. Within each group, the metal lower down the group is more reactive. They have the greatest tendency to lose electrons to form cations.įor the four metals we need to know, those from Group I are more reactive than those from Group II. Group I alkali metals and Group II alkaline earth metals are the most reactive. 
An element's reactivity is its tendency to lose or. The reactivity series is written into the Periodic Table! Highly Reactive Group I and II Metals The reactivities of elements can be predicted by periodic trends (patterns within the Periodic Table.) Let's start with periodic trends. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
